Electronic Trial Master File (eTMF) Software
Features, Development Process, and Investments
In healthcare IT since 2005, ScienceSoft engineers eTMF solutions for CROs, research organizations, pharmaceutical and biotech companies, and health systems. We deliver eTMF software that offers advanced document processing automation capabilities, provides instant visibility into document completeness and timelines, and ensures continuous readiness for regulatory inspections.
Electronic Trial Master File (eTMF) Software at a Glance
Electronic trial master file (eTMF) software provides a secure and structured environment for creating, organizing, and managing clinical trial documentation, tracking the progress of each document in real time, and preparing documentation for health authority inspection.
According to the State of TMF Industry Report 2025, which surveyed 524 TMF professionals across contract research organizations (CROs), pharma, and biotech companies, eTMF adoption is on the rise, with 78% of surveyed organizations using an eTMF system to manage TMF content. 26% of survey respondents cited improving data integrity, traceability, and security as the primary reason for implementing an eTMF system.
Custom eTMF software is often chosen by research organizations that have faced the constraints of ready-made solutions:
- Inability to adapt the TMF structure to specific study SOPs and country-specific regulatory requirements.
- Insufficient scalability of the eTMF solution for handling complex multi-center or multi-country research.
- Limited ability to automate complex documentation management and collaboration workflows, e.g., labor-intensive document classification processes or efficient transfer of data to partners.
- Insufficient analytics capabilities, e.g., for tracking TMF completeness.
- Inability to integrate eTMF with multiple clinical trial systems, including legacy solutions.
Implementation time: from 5 to 10+ months.
Possible integrations for eTMF software: a clinical trial management system (CTMS), electronic data capture (EDC) software, a regulatory electronic document management system (EDMS), document collaboration software, an eSignature tool, and more.
Costs: $80,000–$300,000+, depending on complexity. Answer a few questions about your business needs, and our consultants will get back to you with a custom quote.
Main Capabilities of eTMF Software
Below, ScienceSoft provides an overview of an electronic trial master file system’s functionality that accommodates the key needs of our clients in life science R&D. In real-life projects, every solution has a unique feature set, as it is designed according to the specific requirements of the research organization.
Possible Integrations for eTMF Software
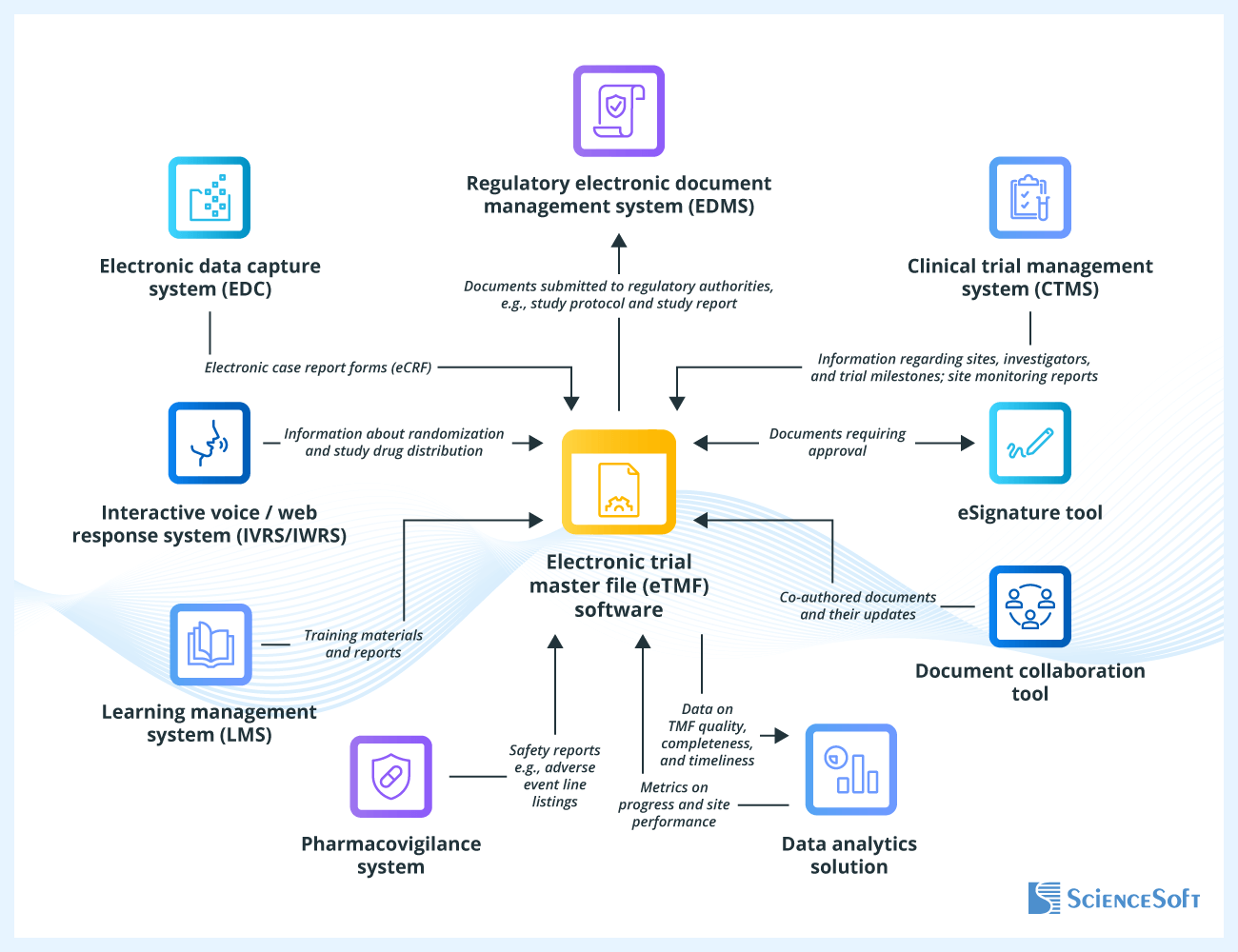
- Clinical trial management system (CTMS) — to obtain data regarding site qualification, initiation, and monitoring, trial milestones, phases, visits, etc.
- Electronic data capture (EDC) system — to collect electronic case report forms (eCRF) with data on patient observations.
- Pharmacovigilance system — to include safety reports, such as adverse event line listings and summary tabulation reports, in the eTMF.
- Regulatory electronic document management system (EDMS) — to transfer documents intended for submission to regulatory authorities.
- Data analytics solution — to retrieve metrics for monitoring site performance, TMF completeness, quality, and timeliness; to forecast document completion timelines and identify potential risks or process bottlenecks; to investigate the root causes of bottlenecks.
Custom eTMF Software Development Process
Below, ScienceSoft’s experts share development steps and best practices to deliver robust electronic master file (eTMF) software while keeping the project’s budget and timelines under control.
1.
Requirement engineering
At this stage, business analysts work with the research organization’s executives, subject matter experts, and future software end-users from the site staff to define the optimal feature set suited to the company's needs.
For example, to address recurring TMF document quality issues, such as missing metadata, a medium-sized research organization might consider implementing a number of more basic features, like pre-configured metadata templates for different document types and mandatory field validation rules when uploading a document.
Meanwhile, a large pharmaceutical company with diverse divisions, external partners, and a high volume of documents might prefer to develop an AI/ML module to automatically check uploaded documents for missing metadata and auto-fill them.
Compliance experts analyze the internal processes of the research organization to create a list of features needed to support compliance with regulations like HIPAA, HITECH, GDPR, FDA 21 CFR Part 11, ICH E6 (R2) GCP, etc.
Planning features that support regulatory compliance requires careful analysis. For example, when planning the design of an electronic document signing service, we follow the FDA 21 CFR Part 11 guidelines, which specify that the reliability of an electronic signature must be equal to that of a signature on a similar paper document.
However, not all of the research organization’s documentation has to comply with this requirement. We work with the client to define the purpose of signatures for each document type. The costly digital signature technology, which ensures the authenticity and integrity of the document's contents, can be reserved for submissions. For internal documents, cheaper alternatives can be used, significantly reducing operational costs, as eTMF documents are signed in bulk.
2.
Architectural design
Software architectures analyze the functional and non-functional requirements documented during business analysis to select a suitable architectural type, define functional modules, and outline interactions between modules.
While designing eTMF software architecture, the specialists ensure it will support all the relevant regulatory requirements. For instance, to plan the implementation of features that will support compliance with FDA 21 CFR Part 11, architects choose logging frameworks for maintaining immutable audit trails and define mechanisms for implementing timestamp accuracy (e.g., NTP, blockchain).
Another essential factor architects consider when designing eTMF architecture and choosing suitable techs is performance, which becomes especially crucial for Phase 3–4 clinical trials, where the eTMF must process tens of thousands of large files.
When choosing integration methods to connect the eTMF solution with existing clinical trial software, architects consider the need to support interoperability standards (HL7 v2/v3, FHIR, USCDI, CCDA) and coding systems (SNOMED CT, ICD-10, CPT, LOINC, RxNorm, MedDRA) to enable proper clinical data integration. For example, to enable real-time access to visit and patient records for the purpose of eTMF document tracking and classification, FHIR APIs are typically developed. When legacy system constraints prevent this, it’s possible to implement integrations via secure file transfer protocol (SFTP) with HL7 data transmission support.
3.
UX and UI design
Designers aim to create role-specific interfaces with easy access to all the automation tools. For example, to reduce site staff's manual data entry during document uploads, designers can ensure that tools like metadata auto-population, document auto-naming, and placeholder auto-generation are readily available directly in the upload interface.
Another important task is to make navigation convenient and the interface elements intuitive. To achieve this, designers can implement breadcrumbs navigation to let users easily track their position within the eTMF and side navigation with collapsible sections to let them view only the areas relevant to the current task. To gather feedback and make adjustments based on user needs and pains, designers may also involve future software users in usability testing during the prototyping stage.
4.
Development and testing
At this point, developers code user interfaces, the back end of eTMF software (including advanced modules like the AI model for TMF document classification), and set up data storage.
Testing usually happens in parallel with development to identify and fix issues as quickly as possible. Solutions for clinical research, which are subject to strict regulatory requirements, demand particularly rigorous security testing. Another major priority is integration testing, as it’s necessary to ensure smooth data flows and prevent delays in uploading or updating large files in TMF.
5.
Deployment and support
Before the solution goes live, developers run a pre-launch compliance audit to double-check the software's compliance with regulations. After the launch, they monitor the solution’s performance, resolve any remaining issues, and organize a knowledge transfer to the research organization’s support engineers, who will handle further eTMF software support and maintenance.
How Much Does It Cost to Develop a Custom eTMF Software?
The costs for building a custom electronic trial master file software vary from $80,000 to over $300,000. The major cost factors include the number of integrations and the need for advanced analytics and AI capabilities.
Here’s an approximate estimate of the costs for building basic, standard, and advanced eTMF solutions, along with a summary of what they offer.
Basic
From $80,000
Main capabilities:
- Document uploading, versioning, viewing, filtering, searching, and exporting.
- Task tracker.
- Basic automated workflows for task assignment.
- Basic TMF readiness tracking.
- TMF archiving.
- Integration with EDC and CTMS.
Standard
From $150,000
In addition to the basic features:
- A wizard for eTMF study set-up.
- Automated workflows for document co-authoring, QC reviewing, and approval.
- Analytics for TMF completeness, quality, and timeliness metrics.
- Integration with regulatory EDMS.
Advanced
From $300,000
In addition to the standard features:
- AI-driven document classification.
- AI-assisted QC reviewing.
- Integration with 4+ systems (e.g., EDC, CTMS, regulatory EDMS, CRM, and LMS).
- Predictive analytics (e.g., to forecast TMF timelines).
Learn the cost of your eTMF software
ScienceSoft's experts are ready to provide a quote for your specific case.
Why Choose ScienceSoft for Your eTMF Project
- Since 2005 in healthcare IT.
- 150+ successful projects in the domain.
- Experience in meeting GCP, FDA, HIPAA, HITECH, GDPR, and 21st Century Cures Act requirements.
- Proficiency in interoperability standards (FHIR, HL7 v2/v3, USCDI, CCDA) and major medical coding systems (ICD-10, CPT, SNOMED CT, LOINC, RxNorm).
- ISO 13485- and ISO 9001-certified quality management, ISO 27001-certified information security management.
Our awards and partnerships
Featured among Healthcare IT Services Leaders in the 2022 and 2024 SPARK Matrix
Recognized for Healthcare Technology Leadership by Frost & Sullivan in 2023 and 2025
Named among America’s Fastest-Growing Companies by Financial Times, 4 years in a row

Top Healthcare IT Developer and Advisor by Black Book™ survey 2023
Recognized by Health Tech Newspaper awards for the third time (2022, 2023, 2025)

Named to The Healthcare Technology Report’s Top 25 Healthcare Software Companies of 2025
ISO 13485-certified quality management system
ISO 27001-certified security management system




